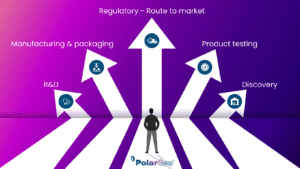
As a key player in the medical field there is a strong focus on innovation and improving patient outcomes but there are many stages that can impact the way you bring your device to market, and in turn have direct results on its success. In the process of developing a medical device there are multiple stages of design and development and a Product Developer may seek guidance from an experienced contract manufacturer with one or more of these stages. As a leading contract manufacturer in the medical device industry, PolarSeal offers a wide range of flexible material converting solutions that enable us to guide you through the entire development process, clearing the pathway for your route to market.

DESIGN
By the time you are ready to speak to a contract canufacturer you will already have a concept, intended market and performance requirements. This initial design stage, is a critical phase in the DEVELOPMENT PROCESS; It involves conceptualizing, planning, and specifying the details of the device’s structure, functionality, and features, so it’s important that a contract manufacturers experts can guide you through the process of developing your design with maximum efficiency, with detailed engineering work to create the technical specifications, and diagrams necessary for converting & manufacturing. So, whether it’s evaluating a new design, or enhancing an existing product PolarSeal can work with you to uncover solutions and accelerate your product and go to market strategy. PolarSeal’s Project Managers provide you with the applicable material samples and their performance against your application as well as the best converting process to achieve your manufacturing goals.
r&d and prototyping
Creating physical or virtual prototypes to validate design concepts, functionality, and ergonomics is another key stage of the process. This may involve 3D printing, CAD (Computer-Aided Design), utilizing a plotter cutter, working with your CMO during this stage ensures that iterations and design amendments do not heavily impact the cost, timeline or manufacturing process of your design, ensuring we can refine the design based on feedback from testing and validation. PolarSeal offer onsite rapid medical device prototyping which allows us to understand your medical device, gaining clear understanding of its intended use within the market and establish its key performance requirements, our R&D experts can bring your product to life and aid you in the process of material and adhesive selection furthermore, PolarSeal can provide sample runs using our rotary converting capabilities This enables us to ensure there is a seamless transition from prototype through to full manufacturing runs.
material selection
Our longstanding relationships with material suppliers and manufacturers enable a medical device designer to take advantage of a wide range of industry giants, access material samples and tap into knowledge unrivalled by other CMOs, choosing appropriate materials that are safe, durable, and compatible with the device’s intended use. Considerations include biocompatibility, sterility, and material strength. PolarSeal’s undisturbed supply chain allows you to focus on the finer details while we coordinate timelines and supply chains on your behalf.
certification
We are proud to be able to perform in-house testing to demonstrate device reliability and safety, ensuring the selected materials and adhesives are the right combination for your application and right for the intended end user, we support you through this process ensuring the stringent requirements for regulatory validation are met. There are multiple routes to market for medical device companies, and PolarSeal assists in this process helping you with classification, document preparation and final submission, gain access to our Regulatory and Quality specialists who will nurture your product through the complex landscape of validation. Verification confirms that the design meets the specified criteria, while validation confirms that it meets the user’s needs and intended use. “Given the requirements on patient safety and device performance, certification requirements and their associated challenges have the potential to cause crucial delays in bringing your product to launch. PolarSeal’s quality and regulatory experts will work with you to implement a timely and efficient route to market and regulatory compliance.”- Christopher Mallett, Quality Manager
manufacture
Manufacturing a medical device demands strict adherence to quality standards, regulatory requirements, and attention to detail. It requires a well-coordinated effort among cross-functional teams, including engineers, quality assurance professionals, production operators, and regulatory experts. PolarSeal will develop a production plan that outlines the sequence of operations, resource allocation, and timeline for your device manufacturing, consider factors like batch sizes, lead times, and resource availability and coordinate production across our huge range of validated capabilities, whether its tight tolerance die cuts, lamination of substrates or roll to roll flexographic printing our experienced operators can cater to your bespoke requirements to streamline the manufacture of your design. “PolarSeal are constantly investing in the training of our teams and development of our processes to deliver efficient and cost-effective solutions for all our customers.”– Simon Bowden, Operations Manager
packaging
Medical device packaging serves several crucial functions that are integral to the safety, efficacy, and usability of the device and transport contamination can be a big concern for medical device designers, that’s why we offer in-house packing as a key capability at PolarSeal. Our pouching packaging machines provide high seal integrity and can work with a wide range of packaging materials, with designs being tailored to your device requirements ensuring moisture and light resistant products. This protection is critical to maintaining the device’s integrity and functionality.

sterilization
Patient care and infection prevention is at the forefront of every medical device designer and manufacturer, and when it comes to some medical device’s STERILIZATION is integral to its market success. PolarSeal can offer a range of sterilization services including Gamma Radiation, E-Beam and EtO.
logistics
On-time and in-full is paramount to meeting your distribution goals. Our long-established logistics networks provide surety of delivery. Additionally, PolarSeal provides CONSIGNMENT STOCK AND WAREHOUSING globally, allowing you to have access to your product when you require.
With a wide range of converting service solutions available it can be difficult to decide the most efficient route to market for your device, but by utilizing the skills, knowledge and experience of a Contract Manufacturer like PolarSeal this pathway can be tailored to suit your medical device. In-house experts can streamline your processes and advise on cost savings.
Read More from PolarSeal
How PolarSeal® Accelerates Your Medical Device’s Journey to Market
Medical device manufacturers face numerous challenges when bringing their innovative products to the market. From regulatory compliance to design and development, each stage requires meticulous

The Importance of Collaboration in Medical Device Manufacturing.
In the dynamic world of healthcare, technological advancements are transforming patient care and treatment methods. One of the driving forces behind these transformations is the

Selecting the optimal wearable adhesive and converting it to perfection
Selecting the correct adhesive for your wearable medical device is crucial to its market success, but with so many adhesives available to the medical market