PolarSeal®
Tapes & Conversions Ltd
We are a fast-moving and highly innovative medical tape and flexible material converter, partnering with you to provide solutions for the co-development of your medical components.
Over the past 40+ years, PolarSeal® has accumulated a wealth of knowledge within a highly skilled team, enabling us to service our clients with the perfect adhesive solution.
We put People first
We Operate with a sense of urgency
We are a Learning organisation
We bring the right Attitude to work
We are Renowned for finding solutions
Our journey and milestones...
1981
Business founded
1994
Original registration to
BSEN/ISO 9001 Standard
1999
First Class 10,000 Cleanroom brought on stream
2006
Certification to ISO13485:2003 Medical Device Standard
2007
2nd ISO7 Cleanroom (Class 10,000) brought on stream
2009
3rd ISO7 Cleanroom (Class10,000) brought on stream
2010
7th Servo Converting machine ordered
2012
Acquisition of a new 36,000ft2 production facility in Suffolk
2013
Registration with FDA
2014
Certification to d. MDD 93/42/EEC
2015
Patent awarded for woundcare product
and manufacturing process
2016
30th Production line validated
2017
4,000 square foot clean production area
2018
4th 2,000 square foot cleanroom installed
2019
Thermoforming & flow wrapping packing process
added in addition to in-line pouch lines
2020
High volume PPE production lines installed
2021
New warehousing
2022
USA premises
Medical device manufacturing
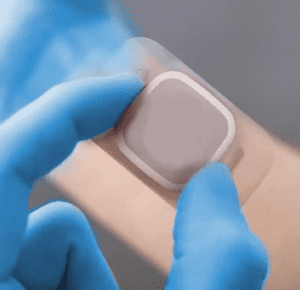
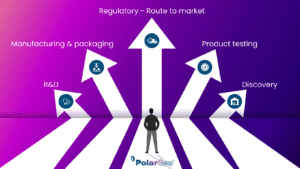
PolarSeal® is solely focused on medical device contract manufacturing and healthcare material conversion.
Operating within our ISO7 cleanroom facilities with ISO13485 accreditations and being FDA Regulated and Audited, PolarSeal® can engineer the perfect adhesive solution.
Our extensive and ever-expanding capabilities allow us to assist global medical device companies in the manufacturing of advanced wound care, ostomy components, wearable device technology, consumer healthcare and so much more.
We understand the growing demand for skin-friendly adhesives and consumer products, and that is why we continuously invest in modern technology and advanced processes to deliver fully qualified products at high speeds and to the ultimate quality standards.
Global manufacturing
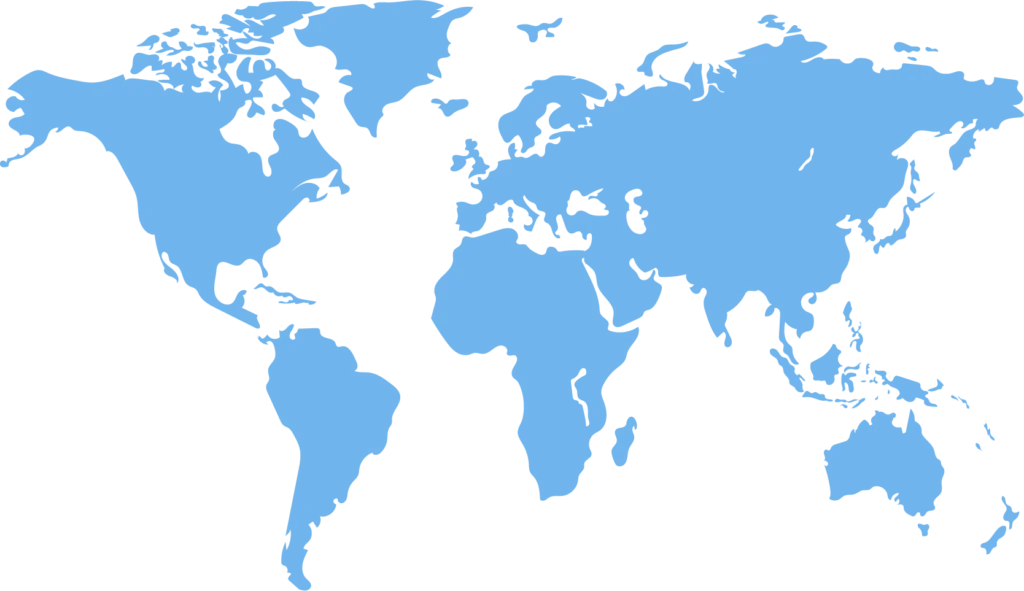
California
New Jersey
Suffolk & Surrey
Company employment statement
As part of the national and international job creation efforts, PolarSeal® strives to continuously provide new opportunities.
Alongside new roles enabled by company growth, PolarSeal® onboards ‘KickStarter’ schemes developed by the Government to assist in the prevention of unemployment and provide futures for the next generation.
Our equal opportunities mission drives us to ensure that we can empower as many individuals as we can through job creation, knowledge transfer, enrichment, and wellness.
Onboarding & developing our people
PolarSeal® undertakes appropriate training, to ensure managers and employees making selection and recruitment decisions do not discriminate, in making decisions.
We adopt a consistent, non-discriminatory approach to the advertising of vacancies where all employees involved in recruitment process will periodically review and offer training on unconscious bias to increase awareness and inclusion during selection criteria, to ensure that they are related to the job requirement and not to discriminate unlawfully.
Promotion and Career development will be made on accomplishment and take account of individual capabilities.