CE marking
Helping launch medical devices into European markets
PolarSeal® make the CE marking process simple.


Certifying medical devices
PolarSeal® is registered and audited by the FDA and by a European Notified Body. PolarSeal® is accredited to CE mark and manufacture our EctoCare range of products which are classified IIb. which means it can CE mark and manufacture classes up to IIb for our EctoCare range of products.
CE marking medical devices is complex as there are multiple criteria to be considered when accurately classifying it, including:
- how long the device is intended to be in continuous use
- whether or not the device is invasive or surgically invasive
- whether the device is implantable or active
- whether or not the device contains a substance, which in its own right is considered to be a medicinal substance and has action ancillary to that of the device.
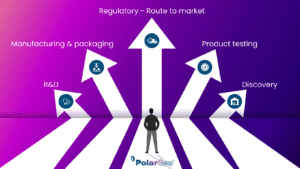
Selecting the appropriate route to conformity assessment
General medical devices: The EU Regulation on Medical Devices 93/42/EEC.
In this context, a medical device is defined as any instrument, apparatus, appliance, software, implant, reagent, material or other article intended by the manufacturer to be used, alone or in combination, for human beings.
Preparation of your technical file
After the classification process is complete, we undertake a rigorous series of tests on your device and comparisons to similar products on the market. The complexity of this testing phase increases in line with the classification of your product.
The results of our testing and other procedures are collated and presented to a Notified Body as a technical file, which is used to qualify the device as a ready-to-sell CE-marked product.
In summary, PolarSeal® makes the whole process easier and propels your product to market.
Preparation of your technical file
The ways in which medical devices manufactured for sale in the UK are regulated may well change as a result of the final BREXIT outcome. We are prepared for all eventualities and remain dedicated to providing you a smooth service.


Get in touch
Click below to make an enquiry to discuss your project requirements in more detail.