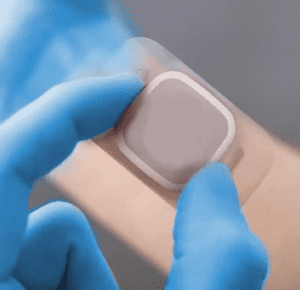
In the dynamic world of healthcare, technological advancements are transforming patient care and treatment methods. One of the driving forces behind these transformations is the collaboration that takes place within the medical device manufacturing process. From design to production, collaboration plays a pivotal role in ensuring the development of safe, effective, and innovative medical devices that ultimately save lives and enhance patients quality of life.
Medical device manufacturing is a complex landscape that requires the harmonious collaboration of various disciplines. Engineering operators, device designers, project managers, R&D Experts, regulatory experts, and supply chain professionals all come together, each contributing their unique expertise. The multidisciplinary nature of this collaboration ensures that medical devices are not only technologically advanced but also clinically relevant and compliant with regulatory standards across a multitude of market.
Operators work hand in hand with quality experts to understand the intricacies of the device, enabling them to produce a device to the highest standards. Regulatory experts ensure that the device meets the stringent guidelines set by organizations like the FDA, EMA & MHRA guaranteeing its safety and efficacy. This interplay of skills results in devices that are not only groundbreaking but also trusted by the medical community.
Sharing Knowledge and Insights
Collaboration accelerates innovation by fostering the exchange of knowledge and insights. When professionals from different backgrounds collaborate, they bring diverse perspectives to the table. This diversity fuels creativity and problem-solving, leading to the development of novel solutions that may not have been possible through isolated efforts.
Navigating Regulatory Landscapes: Compliance and Safety
In the medical field, ensuring patient safety is paramount. Collaborative efforts are vital in navigating the intricate regulatory landscapes that govern medical device manufacturing. Regulatory experts work alongside engineers and clinicians to interpret and adhere to regulations, ensuring that devices are not only innovative but also compliant with industry standards. By collaborating early and consistently with regulatory professionals, designers and manufacturers can identify potential hurdles and compliance issues before they escalate. This proactive approach streamlines the development process, reducing the risk of costly delays and ensuring that devices reach the market efficiently, without compromising on safety.
Supply Chain Efficiency: Seamless Integration
Collaboration extends beyond the confines of the design and development stages. It seamlessly integrates into the supply chain, a critical aspect of medical device manufacturing. Supply chain professionals collaborate with various stakeholders, including suppliers and manufacturers, to ensure the timely and cost-effective delivery of components and materials. Effective collaboration in the supply chain reduces the likelihood of shortages or production delays, unforeseen costs and rapid problem solving. By fostering strong relationships with suppliers, manufacturers can respond swiftly to market demands and unforeseen challenges, guaranteeing a steady flow of devices to healthcare providers.
The Patient-Centric Approach: Improved Outcomes
At the heart of medical device manufacturing collaboration is a patient-centric approach. The collaboration ensures that devices are designed not only with cutting-edge technology but also with the end-user—the patient—in mind. This patient-centric collaboration extends to post-market surveillance as well. Healthcare professionals collaborate with manufacturers to gather real-world data on device performance and patient outcomes. This information is invaluable in fine-tuning devices and addressing any issues that may arise, ensuring continuous improvement in device quality and patient care.
By working collaboratively to manufacture your medical device, your driving innovation, ensuring compliance, and placing patient well-being at the forefront. The synergy of expertise from diverse disciplines paves the way for breakthroughs that revolutionize healthcare practices. In a field where precision and safety are paramount, collaboration is not just a strategy; it’s the heartbeat of progress, ensuring that every medical device that reaches the hands of a healthcare provider is a testament to the power of working together.
Read More from PolarSeal

7 complications when outsourcing to multiple vendors
The service offering of outsourcing your manufacturing process is a common practice within medical device manufacturing, by outsourcing the manufacture of your product you free

What makes our Leadership Team unique?
Family founded businesses hold a special place in the economic landscape, combining the strength of kinship with entrepreneurial spirit, PolarSeal possess a distinct character that
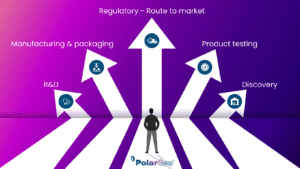
Route to market
Route to market for medical devices Quality and Regulatory getting you market ready PolarSeal® equips you with everything you need to take your medical device