Avery Dennison
MED 5556H
Hydrocolloid
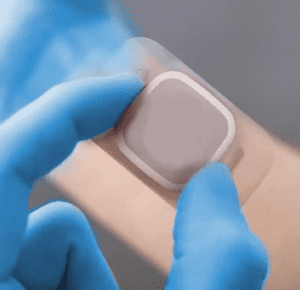
MED 5556H is a single-coated, transparent, polyurethane film containing an integrated hydrocolloid formulation. This product’s absorbent adhesive is designed to limit break down upon saturation, to assist in creating optimal skin and wound healing conditions, and has a high fluid handling capacity.
Typical Application
Designed for wound care, ostomy flange applications and fixation of medical devices.
Features and Benefits
- Conformable
- Integrated hydrocolloid
- High absorption rate
- Moisture resistant
- Printable
- Die-cuttable
- Gamma sterilizable
|
Physical Properties (Not intended as a specification.)
|
Test Method**
|
Typical Values*
|
|---|---|---|
|
Peel Adhesion on Polyethylene (PE)
|
TDS-01
|
0.4 lbf/in 1.9 N/25 mm
|
|
Peel Adhesion on Stainless Steel (SS)
|
TDS-04
|
3.8 lbf/in 16.5 N/25 mm
|
|
Liner Release
|
TDS-06
|
2.7 oz/in 75 g/25 mm
|
|
Reverse Tack
|
TDS-09
|
7.4 lbf/in 32 N/25 mm
|
|
Static Shear
|
TDS-14
|
450 min
|
|
MVTR
|
TDS-17
|
27 g/100 in2 per hr 415 g/m² per 24 hr
|
|
Static Absorption
|
TDS-17
|
455 g/100 in2 per hr 7060 g/m² per 24 hr
|
|
Product Construction
|
Typical Values*
|
Description
|
|---|---|---|
|
Carrier Thickness
|
2.8 mils 70 µm
|
Transparent polyurethane film
|
|
Adhesive Thickness
|
30 mils 750 µm
|
Hydrocolloid designed for medical applications
|
|
Release Liner Thickness
|
2.6 mils 67 µm
|
Paper liner
|
MVTR, moisture vapor transmission rate.
*Refer to product specifications for material acceptance limits.
**Test method information available upon request.
STORAGE AND SHELF LIFE
Two years when stored at 23°C (73°F), 50% relative humidity, out of direct sunlight, in original packaging.