Avery Dennison
MED 5094H
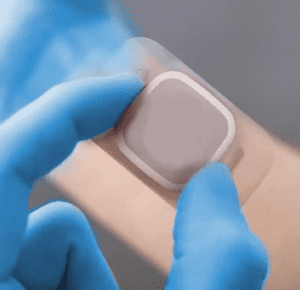
Single-Coated Film Hydrocolloid
MED 5094H is a transparent, coextruded EVA-PVDC-EVA film containing an advanced and integrated hydrocolloid formulation. This product’s absorbent adhesive is designed to not break down upon saturation, provides a low profile, assists in creating optimal skin and wound healing conditions and has a high fluid handling capacity.
Typical Application
Designed for wound care and fixation of medical devices.
Features and Benefits
- Conformable
- Integrated hydrocolloid
- High absorption rate
- Moisture resistant
- Die-cuttable
- Gamma sterilizable
|
Physical Properties (Not intended as a specification.)
|
Test Method**
|
Typical Values*
|
|---|---|---|
|
Peel Adhesion on Polyethylene (PE)
|
TDS-02
|
0.5 lbf/in 2.2 N/25 mm
|
|
Peel Adhesion on Stainless Steel (SS)
|
TDS-04
|
1.1 lbf/in 4.7 N/25 mm
|
|
Liner Release
|
TDS-06
|
2.5 oz/in 42 g/25 mm
|
|
Static Shear
|
TDS-14
|
1560 min
|
|
Reverse Tack
|
TDS-09
|
3.5 lbf/in 15 N/25 mm
|
|
MVTR
|
TDS-17
|
3 [g/100in2 -24 hours] 50 [g/m2 -24 hours]
|
|
Static Absorption
|
TDS-17
|
221 [g/100in2 -24 hours] 3430 [g/m2 -24 hours]
|
|
Product Construction
|
Typical Values*
|
Description
|
|---|---|---|
|
Carrier Thickness
|
3.7 mils 95 μm
|
Transparent co-extruded EVA-PVDC-EVA film
|
|
Adhesive Thickness
|
12 mils 300 μm
|
Hydrocolloid designed for medical applications
|
|
Release Liner Thickness
|
3.4 mils 85 µm
|
White super-calendered glassine
|
*Refer to product specifications for material acceptance limits. **Test method information available upon request.
STORAGE AND SHELF LIFE
Two years when stored at 23°C (73°F), 50% relative humidity, out of direct sunlight, in original packaging.