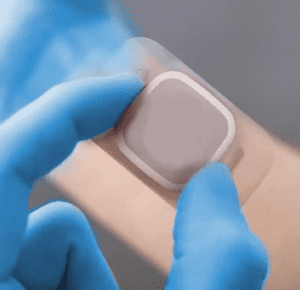
So, you’ve chosen your converting partner, but what can you expect when working with a flexible material converter for medical devices? It is important to establish a clear project timeline, with achievable milestones and direct communication channels to ensure the most efficient management of your project. Your converter should have experience and expertise working with medical-grade materials and be familiar with the regulations and guidelines set by the relevant regulatory bodies. They should have a quality management system (QMS) in place and can ensure, if required that materials used are sterile and free from contaminants, and that the finished product meets the required specifications. In addition to that, it is important for the flexible material converter to have experience in different converting capabilities and technologies, such as lamination, die cutting, and material slitting. This allows them to provide a variety of options for the customer, and to recommend the best solution for a particular project.
The Key Stages of your project
In the initial stages of the project, you may provide your converter with detailed drawings, commonly referred to as BOM or ‘bill of material’, product samples and prototypes for review and feedback, alternatively your converter can work with you collaboratively on design, material selection and can provide you with any necessary regulatory assistance. This allows the customer and converter to make any necessary changes or adjustments before the final product is produced and ensures the final product meets the customer’s requirements and expectations. During this process it’s key that your converter sets realistic timelines and provides updates at regular intervals, as well as communicate openly around any challenges that may arise. This may involve material or tooling lead times and where the importance of rigid supply chain management process is paramount.
Once your final BOM has been signed off, your converter will move forward with your design ensuring the necessary materials, tooling and part numbers are registered and on site ready to begin production. During the manufacturing process, the converter should provide regular updates and progress reports and should be available for any questions or concerns that may arise. Your converter should have a robust testing and quality control process in place to ensure that the final product meets all specifications and regulatory requirements.
Upon manufacture your converter is responsible for ensuring that the final product is packaged as per any agreed conditions, and that those conditions can be maintained during shipment. Your converters storage and transportation capabilities should not be overlooked, it’s important to have reliable logistics to ensure that the finished product is shipped to the customer in a timely manner.
In conclusion, when working with a flexible material converter for your medical device, it is important to have clear communication, a detailed understanding of the project requirements, and a comprehensive quality management system in place. The converter should be experienced in working with medical-grade materials, familiar with regulatory requirements, and have a range of converting capabilities and technologies. They should provide regular updates, be available for questions and concerns, and have a reliable logistics and supply chain management process in place.
PolarSeal can offer services from initial concept through to supply chain management. Find are more here