From manufacturing to legal practices to schools to book publishers, everyone publicises the quality of their products or services. But what is the concept of ‘quality’, and what does it mean, particularly when the quality of your product can affect the health of its user?
It is important to note that in the medical device manufacturing industry, quality is a very real, quantifiable concept. Since biomedical products have a direct impact on consumer health, there are a careful set of guidelines that inform how quality is controlled. These are created and maintained by the ISO 13485 Quality Standard and FDA regulations. These standards regulate a baseline quality control to which all wound care product manufacturers must adhere.
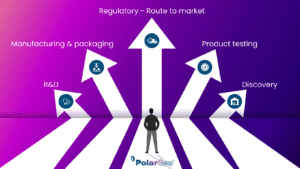
These regulations exist not simply to inform the physical process of manufacturing, but also to standardise the design, development, prototyping, creation, distribution, installation, and servicing within the medical device industry. Having this kind of regulation is helpful as it creates a set standard in a sector that has very real consequences on the health of the general public and ensures the safety and high standards of the products produced.
While it is important to have this set of overarching guidelines in such a critical industry, PolarSeal takes the concept of ‘quality’ beyond what it means on paper. While we adhere to and are committed to the ISO 13845 regulations, we don’t believe that quality begins and ends there. PolarSeal is, first and foremost, a business, and good businesses thrive on providing people with a reliable product or service, in a predictable time frame, and at a consistent pace. In short, good businesses must produce ‘quality’ products and services to retain their status as good businesses.
We don’t simply see ‘quality’ as maintaining the baseline standards of our industry – quality is something that a client understands when they use your product and or service. It is in the dedication and care put into every facet of manufacturing. It is in fostering and maintaining good working relationships with suppliers and distributors. It is in effective communication both internally and externally. Truly caring about what we do leads to quality being recognised in every aspect of our business.
Our commitment to quality is such that we built our entire business around it. Our PolarSeal Quality Policy and Management System is central to our entire existence as a flexible material converter. We have defined, explained, and quantified what we believe quality is when it comes to our service, and every decision we take is taken with this well-defined version of quality in mind.
Our two-pronged approach, adhering to external legal quality control as well as imposing internal quality control structures, allows us to maintain exceptional standards of high-quality continued operations in the medical development field. To learn more about how our quality control can help develop and produce your product, contact us today.