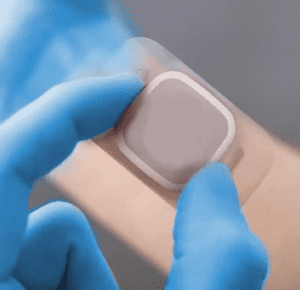
To put it simply, medical device manufacturing refers to a broad range of equipment, tools and devices that are produced for various applications in healthcare.
The medical device manufacturing industry is comprised of a wide range of sectors ranging from diagnostic and surgical devices to cardiovascular devices, diabetes care, and more.
Each sector is responsible for producing equipment, tools and devices that play key roles in the healthcare industry.

What is a medical device?
A medical device generally refers to a healthcare tool or piece of equipment that is used for surgical applications, patient care, urgent care or other medical applications.
“Medical devices” can refer to simple fixation devices and instruments as well as complex diagnostic apparatus and medical monitoring devices.
Types of medical devices
Let’s find out more about some of the types of medical devices that are manufactured for various applications in the healthcare industry.
Whether you’re a patient or practitioner, you may already be quite familiar with a few of the devices listed below.
- Diagnostic apparatus
- Medical monitoring devices
- Orthopaedics
- Orthotics
- Prosthetics
- Surgical instruments
- Implants
- Surgical drapes
- Surgical instruments and technology
- IV equipment
- Fixation devices
- Urology devices
- Neuromodulation devices
- Dental instruments
- Wound care & dressings

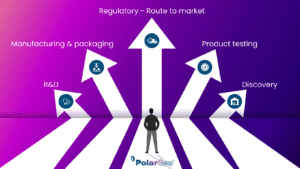
How does manufacturing medical devices work?
Medical device manufacturing is very dependent on the requirements of your devices and their intended medical applications.
Medical device manufacturers design and produce devices with a series of tools in order to provide specific functionality and produce a visual feel that aligns with their client’s expectations.
This requires the use of a range of traditional machinery and production methods often with the added complication of unusual shapes and parts as well as needing to be produced under sterile conditions.
Due to the complexity of these processes, it is often best to outsource the manufacturing of medical devices to expert contract manufacturers.

What does ‘contract medical device manufacturing’ mean?
Contract medical device manufacturing is when the design and/or production of certain components or the whole medical device is outsourced to an external contractor – usually an expert manufacturer with experience, equipment, and a licensed facility with sterile production capabilities.
Medical manufacturing companies often specialise in a particular type of medical manufacturing but others may have more extensive medical manufacturing capabilities.
Important considerations for medical device manufacturing
Medical devices are designed for very specific intended applications, some of which mean they need to be attached to other equipment, or the human body. Depending on how your device is going to be used, you’ll have specific requirements in mind.
Examples of these facets might include durability and strength of materials, waterproofing to guard against sweat and other fluids, and other requirements depending on the application.
PolarSeal works collaboratively with specialists to research, design and develop innovative medical products that match their application. We also improve existing products – defining new ways to reduce costs, increase efficiency, improve effective application and more. Find out more by reading about our R&D and Prototyping Process.
1.Usability / ease-of-use
As medical devices are designed for particular medical applications, their ease of use is paramount to their success and longevity.
In order to ensure that your device delivers its full intended purpose and that it is competitive toward similar products that might serve a similar function, device usability is very important.
2. Movement/travel
Many medical devices are fixed to the body or other equipment/tools and this needs to be taken into account during ideation, design and production.
Particularly for devices that are affixed to the body, regular or limited activities as well as the movement of specific limbs or joints a device may be attached to should be considered.
The selection of specific adhesive is an important consideration when it comes to the movement involved during a medical device’s application.
Adhesives will be selected depending on the material/surface to which they are bound, such as skin or another component. The intended wear time, or application time, for a medical device also needs to be considered. The adhesive chosen may need to be able to withstand bodily fluids, specific temperatures and levels of humidity, as well as other external stresses/influences.
3. Regulations and compliance: design and certification
Medical devices need to be designed in accordance with strict regulatory and manufacturing standards in order to be used for their desired medical application. These regulations are in place to ensure safety and accountability for users of the device.
Depending on the region where the device is used, medical devices may need to adhere to specific policies set out within the EU or the Americas and be certified before distribution of a device/medical product.
We consistently uphold and maintain the fullest quality commitment to globally recognised standards and regulations for manufacturing medical devices, including ISO 13485:2016, MDD 93/42/EEC, MDR regulation 2017/745, ISO 7 Cleanrooms to 14644-1. We are also FDA Registered and Audited.
4. Testing/validation
Medical devices are subjected to stringent testing. This is to ensure their efficacy for intended medical applications, their safety for use on/with patients and practitioners, and address other areas of concern that may be uncovered through thorough testing.
PolarSeal also provides an unbroken commitment to quality and attention to detail, and we’ve successfully built long-standing confidence by producing premium quality, compliant medical devices.
The PolarSeal Quality Policy and Management System includes a commitment to:
- Compliance with all applicable laws and regulations
- The concept of continual development
- Effective communication of quality objectives
- Taking care of employees and visitors
- Working closely with customers and suppliers
- Adopting forward-looking business decisions
- Training staff in the needs of quality management
- Maintaining a dual-site manufacturing capability

Partner with a high-quality (UK & US) medical device manufacturer
We have experience assisting all kinds of companies in healthcare industries around the world with their medical manufacturing needs. We are experts at designing and producing medical devices in accordance with relevant region-specific manufacturing standards and policies.
Our manufacturing capabilities are extensive and we’re ready to meet your exact production requirements. We work with you to ensure you’re ready to enter the market with your medical device that meets the strictest manufacturing standards in the industry.
Many of our clients are based in the UK, EU, and North America, and we design and produce medical products in alignment with specific policies and guidelines depending on your region(s) of operations.
Contact us to find out more about medical device manufacturing today!